1. Abacavir
Nomenclature
CAS number: 136470-78-5
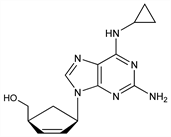
(1S,4R)-4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol; ()-cis-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol; 1592U89. C14H18N6O; mol wt 286.33.
C 58.73%, H 6.34%, N 29.35%, O 5.59%.
Description and references
Nucleoside reverse transcriptase inhibitor (NRTI). Prepn: S. M. Daluge, EP 349242 (1990 to Wellcome Found.); idem, US 5034394 (1991 to Burroughs Wellcome). Asymmetric synthesis: M. T. Crimmins, B. W. King, J. Org. Chem. 61, 4192 (1996) DOI PubMed. Pharmacology and biological profile: S. M. Daluge et al., Antimicrob. Agents Chemother. 41, 1082 (1997) PubMed. Review of antiviral activity and clinical evaluations: R. H. Foster, D. Faulds, Drugs 55, 729-736 (1998) DOI PubMed. Clinical trial of triple nucleoside regimen in HIV patients: S. Staszewski et al., J. Am. Med. Assoc. 285, 1155 (2001) DOI PubMed.
Properties
White solid foam from acetonitrile, mp 165°. uv max (pH 1): 296, 255 nm (ε 14000, 10700); uv max (pH 7): 284, 259 nm (ε 15900, 9200); uv max (pH 13): 284, 259 nm (ε 15800, 9100). [α]D20 59.7°; [α]43620 127.8°; [α]36520 218.1° (c = 0.15 in methanol). Log P (1-octanol/0.1M sodium phosphate): 1.22 ±0.03 (pH 7.4). pKa 5.01. Soly in water (25°): >80 mM (pH 7).Derivative
Sulfate.Nomenclature
CAS number: 188062-50-2
Ziagen (GSK). (C14H18N6O)2.H2SO4; mol wt 670.74.
C 50.14%, H 5.71%, N 25.06%, O 14.31%, S 4.78%.
Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides