2. Abamectin
Nomenclature
Description and references
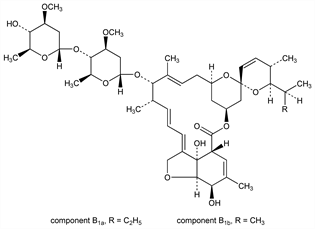
Mixture of avermectins, q.v., containing at least 80% of avermectin B1a ( C48H72O14 ) and not more than 20% of avermectin B1b ( C47H70O14 ). Isoln from Streptomyces avermitilis: G. Albers-Schnberg et al., DE 2717040; eidem, US 4310519 (1977, 1982 both to Merck & Co.). Separation of components: T. W. Miller et al., Antimicrob. Agents Chemother. 15, 368 (1979) PubMed; by semi-preparative HPLC: C. C. Ku et al., J. Liq. Chromatogr. 7, 2905 (1984) DOI. Structure determn: G. Albers-Schnberg et al., J. Am. Chem. Soc. 103, 4216 (1981) DOI. Absolute configuration: J. P. Springer et al., ibid. 4221 DOI. Partial synthesis of B1a: K. C. Nicolaou et al., ibid. 106, 4189 (1984) DOI. Total synthesis: S. Hanessian et al., ibid. 108, 2776 (1986) DOI. Anthelmintic activity: L. S. Blair, W. C. Campbell, J. Parasitol. 64, 1032 (1978) DOI PubMed; J. R. Egerton et al., Antimicrob. Agents Chemother. 15, 372 (1979) PubMed; K. S. Todd et al., Am. J. Vet. Res. 45, 976 (1984) PubMed. Pesticidal activity: I. Putter et al., Experientia 37, 963 (1981) DOI; R. A. Dybas, A. St. J. Green, Proc. Br. Crop Prot. Conf. - Pests Dis. 1984, 947. Control of red imported fire ants: J. A. Greenblatt et al., J. Agric. Entomol. 3, 233 (1986). Interaction with GABA receptors: S. S. Pong et al., J. Neurochem. 34, 351 (1980) DOI PubMed; T. N. Mellin et al., Neuropharmacology 22, 89 (1983) DOI PubMed. Receptor binding studies: S. S. Pong, C. C. Wang, ibid. 19, 311 (1980) DOI PubMed; G. Drexler, W. Sieghart, Eur. J. Pharmacol. 99, 269 (1984) DOI PubMed. Fate in soil and plants: D. L. Bull et al., J. Agric. Food Chem. 32, 94 (1984) DOI; H. A. Moye et al., ibid. 35, 859 (1987) DOI; M. S. Maynard et al., ibid. 37, 178, 184 (1989) DOI. Review: J. R. Babu, ACS Symp. Ser. 380, 91-108 (1988). Reviews of modes of action: C. C. Wang, S. S. Pong, Prog. Clin. Biol. Res. 97, 373-395 (1982) PubMed; D. J. Wright, Biochem. Soc. Trans. 15, 65-67 (1987) PubMed; of insecticidal activities and use: L. Strong, T. A. Brown, Bull. Entomol. Res. 77, 357-389 (1987). Book: Ivermectin and Abamectin, W. C. Campbell, Ed. (Springer-Verlag, New York, 1989) 363 pp.