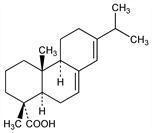
7. Abietic Acid
Nomenclature
CAS number: 514-10-3
(1R,4aR,4bR,10aR)-1,2,3,4,4a,4b,5,6,10,10a-Decahydro-1,4a-dimethyl-7-(1-methylethyl)-1-phenanthrenecarboxylic
acid; 13-isopropylpodocarpa-7,13-dien-15-oic acid; sylvic acid. C20H30O2; mol wt 302.45.
C 79.42%, H 10.00%, O 10.58%.
Description and references
A widely available organic acid, prepared by isomerization of rosin: Harris, Sanderson, Org. Synth. coll. vol. IV, 1 (1963); Fieser, Fieser, The Chemistry of Natural Products Related to Phenanthrene (New York, 3rd ed., 1949). Synthesis from dehydroabietic acid: A. W. Burgstahler, L. W. Worden, J. Am. Chem. Soc. 83, 2587 (1961) DOI; E. Wenkert et al., ibid. 86, 2038 (1964) DOI. Chromatographic study: A. G. Douglas, T. G. Powell, J. Chromatogr. 43, 241 (1969) DOI. Metabolism in rabbits: Y. Asakawa et al., Xenobiotica 16, 753 (1986) PubMed.