18. Acarbose
Nomenclature
Description and references
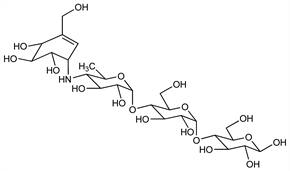
Pseudotetrasaccharide containing an unsaturated cyclitol moiety. An α-glucosidase inhibitor that reduces sugar absorption in the gastrointestinal tract. Isoln from strains of Actinoplanes: W. Frommer et al., DE 2347782; eidem, US 4062950 (1975, 1977 both to Bayer). Total synthesis: S. Ogawa, Y. Shibata, Chem. Commun. 1988, 605; eidem, Carbohydr. Res. 189, 309 (1989) DOI. Biosynthetic studies: U. Degwert et al., J. Antibiot. 40, 855 (1987) PubMed. Glucosidase inhibition studies: D. D. Schmidt et al., Naturwissenschaften 64, 535 (1977) DOI PubMed; W. Puls et al., ibid. 536. Use in treatment of diabetic adults: D. Sailor, G. Roder, Arzneim.-Forsch. 30, 2182 (1980); H. Laube et al., ibid. 1154. Long-term study in sulfonylurea-treated diabetics: H. Vierhapper et al., Diabetologia 20, 586 (1981). Potential use in prophylaxis of dental caries: N. E. Fiehn, D. Moe, Scand. J. Dent. Res. 90, 124 (1982) PubMed. Review of pharmacodynamics, pharmacokinetics and therapeutic potential: S. P. Clissold, C. Edwards, Drugs 35, 214-243 (1988); of cardiovascular benefits in type 2 diabetes: U. Zeymer, Int. J. Cardiol.107, 11-20 (2006) DOI PubMed.
Properties
White to off-white powder. [α]D18 +165° (c = 0.4 in water). Sol in water. pKa 5.1.Therapeutic Category
Antidiabetic.
Therapeutic Category (Veterinary)
Antidiabetic.Keywords
Antidiabetic; α-Glucosidase Inhibitor