19. Acebutolol
Nomenclature
Description and references
Cardioselective β-adrenergic blocker. Prepn: K. R. H. Wooldridge, B. Basil, ZA 6808345; eidem, US 3857952 (1969, 1974 both to May & Baker). Pharmacology: Cuthbert, Owusu-Ankomah, Br. J. Pharmacol. 43, 639 (1971) PubMed; Basil et al., Br. J. Pharmacol. 48, 198 (1973) PubMed; Lewis et al., Br. Heart J. 35, 743 (1973) DOI PubMed. HPLC determn in plasma and urine: M. Piquette-Miller et al., J. Chromatogr. 526, 129 (1990) DOI PubMed. Crystal structure: A. Carpy et al., Acta Crystallogr. B35, 185 (1979). Review of pharmacology and therapeutic efficacy: B. N. Singh et al., Drugs 29, 531-569 (1985) DOI PubMed; G. DeBono et al., Am. Heart J. 109, 1211-1223 (1985) DOI PubMed. Comprehensive description: R. T. Foster, R. A. Carr, Anal. Profiles Drug Subs. 19, 1-26 (1990).
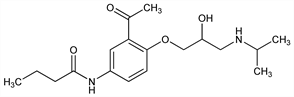
Properties
Crystals, mp 119-123°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from anhydr methanol-anhydr diethyl ether, mp 141-144°. Sol in alcohol, water. Very slightly sol in acetone, methylene chloride. Practically insol in ether. Soly at room temperature (mg/ml): water 200; ethanol 70.Therapeutic Category
Antihypertensive; antianginal; antiarrhythmic (class II).
Keywords
β-Adrenergic Blocker; Antianginal; Antiarrhythmic; Antihypertensive; Aryloxypropanolamine Derivatives