Nomenclature
CAS number: 83-32-9
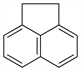
1,2-Dihydroacenaphthylene;
peri-ethylenenaphthalene; 1,8-ethylenenaphthalene.
C
12H
10; mol wt 154.21.
C 93.46%, H 6.54%.
Description and references
Occurs in coal tar. Isoln: Ges. f. Teerverwertung, DE 277110; Chem. Zentralbl. 1914, II, 597. In petroleum residues: Orloff et al., C.A. 31, 28009 (1937). By passing ethylene and benzene or naphthalene though
a red hot tube: Berthelot, Bull. Soc. Chim. [2] 7, 274; 8, 226, 245 (1867).
By heating tetrahydroacenaphthene with sulfur to 180°: Braun et al., Ber. 55, 1694 (1922). From acenaphthenone or acenaphthenequinone by high
pressure hydrogenation in decalin with nickel at 180-240°: Braun,
Bayer, Ber. 59, 921,
923 (1926). From acenaphthenone oxime: Morgan, Stanley, J. Soc. Chem. Ind. London 44,
494T (1925). Review of toxicology and human exposure: Toxicological Profile for Polycyclic Aromatic Hydrocarbons (PB95-264370, 1995) 487 pp.
Properties
Orthorhombic bipyramidal needles from alcohol.
d 1.189. mp 95°. bp 279°. uv spectrum: Seshan, Proc. Indian Acad. Sci. A3, 148
(1936). Factors influencing the uv spectrum: Jones, J. Am. Chem. Soc. 67, 2127 (1945).
Insol in water. One gram dissolves in 31 ml alcohol, 56 ml methanol,
25 ml propanol, 2.5 ml chloroform, 5 ml benzene or toluene. 3.2 g
are sol in 100 ml glacial acetic acid. Forms water-sol, cryst complexes
with desoxycholic acid, containing two molecules of the bile acid
as a rule. The complexes crystallize when concentrated solns of the
proper amount of the components in alcohol or dioxane are allowed
to cool slowly.Use
Dye intermediate; manuf plastics; insecticide; fungicide.
