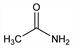
43. Acetamide
Nomenclature
CAS number: 60-35-5
Acetic acid amide. C2H5NO; mol wt 59.07.
C 40.67%, H 8.53%, N 23.71%, O 27.09%.
Description and references
Prepd by fractional distillation of ammonium acetate: Coleman, Alvarado, Org. Synth. coll. vol. I, 3 (2nd ed., 1941); Gattermann-Wieland, Praxis des Organischen Chemikers (40th ed., 1961) p 118; Vogel, Practical Organic Chemistry (3rd ed., 1959) p 401. Prepn from methyl acetate, W. P. Munro et al., US 2106697 (1936 to Calco Chem.); from ethyl acetate, Vogel, op. cit., p 403. Studies of acetamide as an ionizing solvent: Jauder, Winkler, J. Inorg. Nucl. Chem. 9, 24, 32, 39 (1959) DOI. Toxicological study: Weisburger et al., Toxicol. Appl. Pharmacol. 14, 163 (1969) DOI PubMed.