44. Acetamidine
Nomenclature
CAS number: 143-37-3
Ethanimidamide; ethanamidine; α-amino-α-iminoethane; ethenylamidine; acediamine; acetylamidine. C2H6N2; mol wt 58.08.
C 41.36%, H 10.41%, N 48.23%.
Description and references
Prepd by passing HCl into a soln of acetonitrile in abs alcohol, then passing NH3 into the reaction mixture: Pinner, Ber. 16, 1654 (1883); 17, 178 (1884); A. W. Dox, Org. Synth. 8, 1 (1928); by reduction of nitro-2-methylglyoxaline: R. G. Fargher, J. Chem. Soc. 117, 668 (1920) DOI. Prepn from the hydrochloride: I. Crossland, F. S. Grevil, Acta Chem. Scand. B 35, 605 (1981) DOI. Review of chemical behavior and use in syntheses: R. L. Shriner, F. W. Neumann, Chem. Rev. 35, 351-425 (1944) DOI.
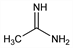
Properties
Colorless needles from methylene chloride, mp 66-67°. uv max: 224 nm (ε 4000). pK1 (25°): 12.1. Has a strong alkaline reaction and on slight warming dissociates into ammonia and acetic acid.Derivative
Hydrochloride.Nomenclature
CAS number: 124-42-5
SN-4455. C2H6N2.HCl; mol wt 94.54.
C 25.41%, H 7.46%, N 29.63%, Cl 37.50%.