47. Acetaminophen
Nomenclature
Description and references
Synthetic non-opiate analgesic. Prepn from p-nitrophenol: Morse, Ber. 11, 232 (1878); Tingle, Williams, Am. Chem. J. 37, 63 (1907); from p-aminophenol: Lumière et al., Bull. Soc. Chim. Fr. [3] 33, 785 (1905); Fierz-David, Kuster, Helv. Chim. Acta 22, 94 (1939); Wilbert, De Angelis, US 2998450 (1961 to Warner-Lambert); Bergmann, DE 453577; Chem. Zentralbl. 1928 I,, 2663; Frdl. 16, 238; from p-hydroxyacetophenone hydrazone: Pearson et al., J. Am. Chem. Soc. 75, 5907 (1953). Evaluation of renal effects: D. P. Sandler et al., N. Engl. J. Med. 320, 1238 (1989) PubMed. Clinical trial in osteoarthritis: A. R. Temple et al., Clin. Ther. 28, 222 (2006) DOI PubMed. Molecular toxicology: P. D. Josephy, Drug Metab. Rev. 37, 581 (2005) DOI PubMed. Comprehensive description: J. E. Fairbrother, Anal. Profiles Drug Subs. 3, 1-109 (1974). Review of pharmacology: B. Ameer, D. J. Greenblatt, Ann. Intern. Med. 87, 202-209 (1977) PubMed. Review of mechanism of hepatotoxicity: L. P. James et al., Drug Metab. Dispos. 31, 1499-1506 (2003) DOI PubMed; of acetaminophen-induced acute liver failure: A. M. Larson et al., Hepatology 42, 1364-1372 (2005) DOI PubMed.
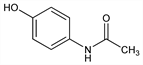
Properties
Large monoclinic prisms from water, mp 169-170.5°. d421 1.293. uv max (ethanol): 250 nm (ε 13800). Freely sol in alcohol. Sol in methanol, ethanol, dimethylformamide, ethylene dichloride, acetone, ethyl acetate, boiling water, 1 N sodium hydroxide. Slightly sol in ether. Very slightly sol in cold water. Practically insol in petr ether, pentane, benzene. LD50 in mice (mg/kg): 338 orally, 500 i.p. See: G. A. Stramer et al., Toxicol. Appl. Pharmacol. 19, 20 (1971) DOI PubMed; D. C. Dahlin, S. D. Nelson, J. Med. Chem. 25, 885 (1982) DOI PubMed.Use
Manuf azo dyes, photographic chemicals.Therapeutic Category
Analgesic; antipyretic.
Therapeutic Category (Veterinary)
Analgesic.Keywords
Analgesic; Non-opioids; Anilides; Antipyretic