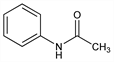
50. Acetanilide
Nomenclature
CAS number: 103-84-4
N-Phenylacetamide; antifebrin; acetylaniline; acetylaminobenzene. C8H9NO; mol wt 135.16.
C 71.09%, H 6.71%, N 10.36%, O 11.84%.
Description and references
p-Aminophenol deriv related to acetaminophen, q.v., with analgesic and antipyretic properties. Usually prepd from aniline and acetic acids: A. I. Vogel, Practical Organic Chemistry (London, 3rd ed., 1959) p 577. From aniline and acetyl chloride: Gattermann-Wieland, Praxis des Organischen Chemikers (Berlin, 40th ed., 1961) p 114. From aniline, acetone and ketene: Hurd, Org. Synth. coll. vol. I (New York, 2nd ed., 1941) p 332. Chemistry: G. C. Derick, J. H. Bornman, J. Am. Chem. Soc. 35, 1269 (1913). Toxicity data: P. K. Smith, W. E. Hambourger, J. Pharmacol. Exp. Ther. 54, (1935). Monograph: Gross, Acetanilid (Hillhouse Press, New Haven, 1946).