Nomenclature
CAS number: 64-19-7
Glacial acetic acid; ethanoic acid; Aci-Jel (Ortho).
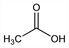
C
2H
4O
2; mol wt 60.05.
C 40.00%, H 6.71%, O 53.29%.
Description and references
Obtained in the destructive distillation of
wood; from acetylene and water, via acetaldehyde by oxidation with
air. Manuf processes: Bhattacharyya,
Sourirajan, J. Appl. Chem. 6, 442 (1956); eidem, ibid. 9, 126 (1959); Elce et al., US 2800504 (1957 to Distillers Co.); Wirth, US 2818428 (1957 to British Petroleum); McKusick and Hoover, US 2940913 and US 2940914 (both 1960 to Du Pont); Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York,
4th ed., 1975) pp 8-15. Toxicity data: H. F. Smyth et al., Arch. Ind.
Hyg. Occup. Med. 4, 119 (1951). Review: F. S. Wagner in Kirk-Othmer
Encyclopedia of Chemical Technology vol. 1 (Wiley-Interscience,
New York, 3rd ed., 1978) pp 124-147.
Properties
Liquid; pungent odor. d16.67 (liq) 1.053; d16.60 (solid) 1.266. d2525 1.049. bp 118°.
mp 16.7°. nD20 1.3718. Flash pt, closed cup: 103°F (39°C). Corrosive, flammable. Contracts slightly on freezing.
Excellent solvent for many organic compounds; also dissolves phosphorus,
sulfur and halogen acids. Miscible with water, alcohol, glycerin,
glycerol, ether, carbon tetrachloride. Practically insol in carbon
disulfide. Weakly ionized in aq solns: pKa 4.74. pH of aq solns 1.0M =
2.4; 0.1M = 2.9; 0.01M = 3.4. LD50 in rats (g/kg): 3.53 orally (Smyth).Incompat. Carbonates, hydroxides, many
oxides, phosphates.
Caution
Ingestion may cause severe corrosion
of mouth and G.I. tract, with vomiting, hematemesis, diarrhea, circulatory
collapse, uremia, death. Chronic exposure may cause erosion of dental
enamel, bronchitis, eye irritation, cf. Patty's Industrial Hygiene and Toxicology vol. 2C, G. D. Clayton, F. E. Clayton, Eds. (Wiley-Interscience,
New York, 3rd ed., 1982) p 4909-4911.Use
Manuf various acetates, acetyl compounds, cellulose
acetate, acetate rayon, plastics and rubber in tanning; as laundry
sour; printing calico and dyeing silk; as acidulant and preservative
in foods; solvent for gums, resins, volatile oils and many other substances.
Widely used in commercial organic syntheses. Aqueous and nonaqueous
acid-base titrations. Trace metal analysis. Pharmaceutic aid (acidifier).
Therapeutic Category (Veterinary)
Vesicant, caustic, destruction of warts.
