Nomenclature
CAS number: 108-24-7
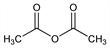
Acetic acid 1,1′-anhydride; acetic oxide; acetyl oxide; ethanoic anhydride.
C
4H
6O
3; mol wt 102.09.
C 47.06%, H 5.92%, O 47.02%.
Description and references
Equivalent to 117.64% acetic acid. Made formerly
from sodium acetate and acetyl or sulfuryl chloride; can be obtained
from acetaldehyde or acetic acid: Faith,
Keyes & Clark's Industrial Chemicals, F. A. Lowenheim,
M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp
16-20. Of industrial importance is also the ketene process, starting
with the thermal decompn of acetone: Schmidlin, Bergmann, Ber. 43, 2821 (1910). Review
of bulk manufacturing process: V. H. Agreda et al., Chemtech 22, 172-181 (1992).
Toxicity study: H. F. Smyth et al., Arch. Ind. Hyg. Occup. Med. 4, 119 (1951).
Review of synthetic uses: D. H. Kim, J.
Heterocycl. Chem. 13, 179-194 (1976); G. B.
Baker et al., J. Pharmacol. Toxicol.
Methods 31, 141-148 (1994).
Properties
Very refractive liquid; strong acetic odor. Flammable, corrosive and combustible. Reacts
with alcohol, oxidizing materials, alkaline materials. Flash pt, closed cup: 120°F (49°C). d415 1.080. mp 73°. bp 139°. nD20 1.3904. Slowly sol
in water, forming acetic acid; with alcohol forms ethyl acetate; sol
in chloroform, ether. LD50 orally in rats: 1.78 g/kg (Smyth).Caution
Potential symptoms of overexposure
are conjunctivitis, lacrimation, corneal edema, opacity and photophobia;
nasal, pharyngeal irritation; cough, dyspnea and bronchitis; skin
burns, vesiculation and sensitization dermatitis. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH
97-140, 1997) p 2.Use
Manuf acetyl compounds, cellulose acetates. As
acetulizer and solvent in examining wool fat, glycerol, fatty and
volatile oils, resins; detection of rosin. Widely used in organic
syntheses, e.g., as dehydrating agent in nitrations, sulfonations
and other reactions where removal of water is necessary. In prepn
of anhydrous acetic acid in nonaqueous titrimetry.
