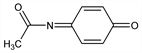
57. Acetimidoquinone
Nomenclature
Description and references
Proposed “ultimate” toxic metabolite of acetaminophen, q.v. Initially prepared by oxidation of acetaminophen with lead tetraacetate and characterized as a Diels-Alder adduct: I. C. Calder et al., J. Med. Chem. 16, 499 (1973) DOI PubMed. Electrochemical generation in buffer: D. J. Miner, P. T. Kissinger, Biochem. Pharmacol. 28, 3285 (1979) DOI PubMed. Synthesis in stable benzene soln and reactivity: I. A. Blair et al., Tetrahedron Lett. 21, 4947 (1980) DOI. Prepn in cryst form, decomposition kinetics, preliminary toxicological studies: D. C. Dahlin, S. D. Nelson, J. Med. Chem. 25, 885 (1982) DOI PubMed. Microsomal reactivity study: G. B. Corcoran, Adv. Exp. Med. Biol. 136B, 1085 (1982) PubMed. Review of acetaminophen-induced hepatotoxicity: J. A. Hinson et al., Life Sci. 29, 107-116 (1981) DOI PubMed.