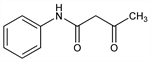
58. Acetoacetanilide
Nomenclature
CAS number: 102-01-2
3-Oxo-N-phenylbutanamide; α-acetylacetanilide; acetoacetic anilide; β-ketobutyranilide. C10H11NO2; mol wt 177.20.
C 67.78%, H 6.26%, N 7.90%, O 18.06%.
Description and references
Prepd by the reaction of ketene dimer with aniline: Chick, Wilsmore, J. Chem. Soc. 1908, 946; Boese, Ind. Eng. Chem. 32, 16 (1940); Williams, Krynitsky, Org. Synth. coll. vol. III, 10 (1955). By the reaction of aniline with ethyl acetoacetate: Knorr, Ann. 26, 69 (1886); Roos, Ber. 21, 624 (1883); Knorr, Reuter, Ber. 27, 1169 (1894). Enol form (unstable) prepd by pouring an alkaline soln of the keto form in cold, dilute H2SO4.