Nomenclature
CAS number: 75-86-5
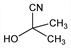
2-Hydroxy-2-methylpropanenitrile; 2-methyllactonitrile; α-hydroxyisobutyronitrile.
C
4H
7NO; mol wt 85.10.
C 56.45%, H 8.29%, N 16.46%, O 18.80%.
Description and references
Prepd by adding acetone to sodium or potassium
cyanide in water and treating with H2SO4 at
below 20°: Welch, Clemo, J. Chem. Soc. 1928, 2629; Cox, Stormont, Org.
Synth. coll. vol. II, 7 (1943). Continuous
production from HCN and aq acetone: G. Barsky, US 2731490 (1956). Toxicity study: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 23, 95 (1962).
Properties
Liquid.
Poisonous. d
425 0.9267; d
419 0.932. mp 19°. bp
15 81°; bp
20 88-90°; bp
23 82°; bp
760 95°.
nD19 1.40002;
nD15 1.3980. Freely sol in water and in the usual organic solvents. Practically
insol in petr ether and carbon disulfide. Decomposes readily to form
hydrogen cyanide,
q.v., which is
highly toxic. LD
50 orally in rats: 0.17 g/kg (Smyth).
Caution
Potential symptoms of overexposure
are irritation of eyes, skin, respiratory system; dizziness, weakness,
headache, confusion, convulsions; liver, kidney injury; pulmonary
edema, asphyxia. See NIOSH Pocket
Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p
4.Use
In preparative organic chemistry for transcyanohydrination,
such as preparing the 17-monocyanohydrin of a 3,17-diketo steroid
by hydrogen cyanide exchange with the reagent,
e.g. Ercoli,
de Ruggieri,
J. Am. Chem. Soc. 75, 650 (1953).
