77. 21-Acetoxypregnenolone
Nomenclature
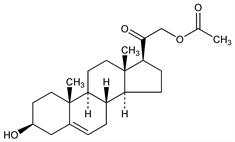
CAS number: 566-78-9
(3β)-21-(Acetyloxy)-3-hydroxypregn-5-en-20-one; 3β,21-dihydroxypregn-5-en-20-one 21-acetate; 5-pregnene-3β,21-diol-20-one 21-acetate; 21-acetoxy-5-pregnen-3-ol-20-one; 3-hydroxy-21-acetoxy-5-pregnen-20-one; prebediolone acetate; A.O.P.; Acetoxanon (Organon); Artisone acetate (Wyeth). C23H34O4; mol wt 374.51.
C 73.76%, H 9.15%, O 17.09%.
Description and references
Intermediate product in the synthesis of desoxycorticosterone acetate: Steiger, Reichstein, Helv. Chim. Acta 20, 1164 (1937). Prepd by reacting 5,6-pregnen-3-ol-20-one with bromine in chloroform and treating the obtained oily bromide with potassium acetate in alcohol: Herloff, Inhoffen, US 2409043 (1946 to Schering).
Properties
Needles from acetone. Becomes opaque at about 80° or on prolonged standing. mp 184-185°. Very slightly sol in ether, in pentane; sol in chloroform, toluene. The esterified keto group is relatively stable to the action of chromic acid in cold and to such reducing agents as potassium iodide or zinc. On heating with aluminum isopropoxide in isopropyl alcohol it is reduced to 5-pregnene-3,20,21-triol.Therapeutic Category
Anti-inflammatory.
Keywords
Glucocorticoid