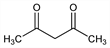
81. Acetylacetone
Nomenclature
CAS number: 123-54-6
2,4-Pentanedione; diacetylmethane. C5H8O2; mol wt 100.12.
C 59.98%, H 8.05%, O 31.96%.
Description and references
Made by the action of sodium on ethyl acetate and acetone or by the action of anhydrous aluminum chloride on acetyl chloride in the presence of an inert solvent: Combes, Compt. Rend. 103, 814 (1886); Ann. Chim. (Paris) 12, 199 (1887); Claisen, Ber. 38, 695 (1905); Hunt, US 2737528 (1956 to Shawinigan Chem.); Georgieff, US 2834811 (1958); Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed, 1961) p 219. Toxicity data: Carpenter, J. Ind. Hyg. Toxicol. 31, 343 (1949).