88. Acetylcysteine
Nomenclature
Description and references
Thiol-containing antioxidant. Prepn: Smith, Gorin, J. Org. Chem. 26, 820 (1961) DOI. Prepn and use in treatment of respiratory diseases: Martin, Waller, US 3184505 (1965 to Mead Johnson). Effect in corneal healing: F. Menna et al., Bull. Mem. Soc. Fr. Opthalmol. 94, 425 (1982); G. Petroutsos et al., Ophthalmic Res. 14, 241 (1982) PubMed. Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971) DOI PubMed. Clinical trial in acetaminophen overdose: M. J. Smilkstein et al., N. Engl. J. Med. 319, 1557 (1988) PubMed; as renal protectant with radiocontrast agents: M. Tepel et al., N.Engl.J.Med. 343, 180 (2000) DOI PubMed. Review: G. R. McKinney, G. M. Sisson, in Pharmacological and Biochemical Properties of Drug Substances vol. 2, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1979) pp 479-488. Review of use in clinical toxicology: R. J. Flanagan, T. J. Meredith, Am. J. Med. 91, Suppl. 3C, 131S-139S (1991).
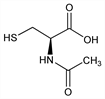
Properties
Crystals from water, mp 109-110°. Slight acetic odor. Freely sol in water, alcohol. Practically insol in chloroform, ether. LD50 orally in rats: 5050 mg/kg (Goldenthal).Therapeutic Category
Mucolytic; corneal vulnerary; antidote (acetaminophen poisoning).
Therapeutic Category (Veterinary)
Expectorant.Keywords
Antidote (Acetaminophen Poisoning); Mucolytic; Vulnerary