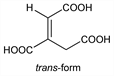
117. Aconitic Acid
Nomenclature
CAS number: 499-12-7
1-Propene-1,2,3-tricarboxylic acid; equisetic acid; citridic acid; achilleic acid. C6H6O6; mol wt 174.11.
C 41.39%, H 3.47%, O 55.14%.
Description and references
Found in leaves and tubers of Aconitum napellus L., Ranunculaceae, in various species of Achillea (Compositae) and Equisetum (Equisetaceae), in beet root, and in sugar cane. Can be prepd commercially from calcium magnesium aconitate recovered from sugar cane juice: McCalip, Seibert, Ind. Eng. Chem. 33, 637 (1941); from molasses: Regna, Bruins, ibid. 48, 1268 (1956). Most of the commercial aconitic acid is, however, manufactured by sulfuric acid dehydration of citric acid: Bruce, Org. Synth. coll. vol. II, p 12 (1943); by using methanesulfonic acid instead of sulfuric: Cranston, US 2727066 (1955 to Daniel F. Kelly). Aconitic acid prepd by any of the above methods has the trans-configuration which is the form described here.