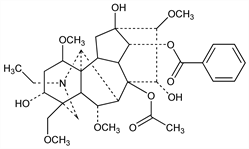
118. Aconitine
Nomenclature
Description and references
Several isomers from Aconitum napellus L., Ranunculaceae and other aconites. Majima et al., Ber. 57, 1456 (1924); Proc. Imp. Acad. Tokyo 5, 415 (1929); Freudenberg, Ber. 69, 1964 (1936); Swanson et al., Aconite, A. Ph. A. Monograph no. 1 (1938); Methods of Analysis, A.O.A.C., 8th ed., 598, 651 (1955). Structure: Wiesner et al., Collect. Czech. Chem. Commun. 28, 2462 (1963); Wiesner et al., Can. J. Chem. 47, 2734 (1969). Stereochemistry: Bachelor et al., Tetrahedron Lett. 1960, no. 10, 1; Gilman, Marion, ibid. 1961, 923; Tsuda, Marion, Can. J. Chem. 41, 1634 (1963); Birnbaum et al., Tetrahedron Lett. 1971, 867. Pharmacology: H. Sato et al., Tohoku J. Exp. Med. 128, 175 (1979). Inotropic effects: P. Honerjger, A. Meissner, Arch. Pharmacol. 322, 49 (1983). Toxicity: Dybing et al., Acta Pharmacol. Toxicol. 7, 337 (1951).
Properties
Hexagonal plates, mp 204°. [α]D +17.3° (chloroform). Aq soln is alkaline to litmus. pK 5.88. One gram dissolves in 2 ml chloroform, 7 ml benzene, 28 ml abs alcohol, 50 ml ether, 3300 ml water. Slightly sol in petr ether. Poisonous. LD50 in mice (mg/kg): 0.166 i.v.; 0.328 i.p.; approx 1 orally (Dybing); also reported as LD50 in mice (mg/kg): 1.8 orally, 0.270 s.c.; 0.380 i.p.; 0.12 i.v. (Sato).Derivative
Hydrobromide hemipentahydrate.Properties
Hexagonal tablets from water, mp 200-207° (sinters at 160°). Amorphous form mp 115-120°. Also crystallizes from ethanol + ether with H2O, mp 206-207°; [α]D 30.8°. Poisonous. Ref: Paech, Tracey, Modern Methods of Plant Analysis vol. IV (Springer-Verlag, Berlin, 1955) p 375.Derivative
Hydrochloride hemipentahydrate.Properties
Crystals, mp 149-153°. mp 194-195° (dry). [α]D 30.9°. Poisonous. Ref: Paech, Tracey.Derivative
Nitrate.Properties
Crystals, mp about 200° (dec). [α]D20 35° (c = 2 in water). Poisonous. One gram dissolves in 10 ml boiling water. Less sol in cold water. Sol in alcohol.Caution
Potential symptoms of overexposure are nausea, vomiting, diarrhea due to CNS stimulation; restlessness, ataxia, vertigo, slow and dyspneic breathing, hypothermia, convulsions. Direct contact may cause warm tingling sensation with subsequent numbness on mouth and mucous membranes. See Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984) Section II, p 249.Use
Used in producing heart arrhythmia in experimental animals: Boyadzhiev, C.A. 73, 86256e (1970).Therapeutic Category
Has been used topically in neuralgia.