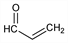Nomenclature
CAS number: 107-02-8
2-Propenal; acrylic aldehyde; acrylaldehyde; acraldehyde; Aqualin (Shell); Magnacide (Baker).
C
3H
4O; mol wt 56.06.
C 64.27%, H 7.19%, O 28.54%.
Description and references
Prepd industrially by passing glycerol vapors
over magnesium sulfate heated to 330-340°. Lab prepn by heating a
mixture of anhydr glycerol, acid potassium sulfate and potassium sulfate
in the presence of a small amount of hydroquinone and distilling in
the dark: H. Adkins, W. H. Hartung, Org.
Synth. coll. vol. I, 15 (1941). Formation
from glycerol by the action of B. amaracrylus: Voisenet, Compt. Rend. 188, 941, 1271 (1929);
by B. welchii: Humphreys, J. Infect.
Dis. 35, 282; Chem.
Zentralbl. 1925, II, 309. Toxicity study:
H. F. Smyth et al., Arch. Ind.
Hyg. Occup. Med. 4, 119 (1951). Review: L. G. Hess et al., in Kirk-Othmer
Encyclopedia of Chemical Technology vol. 1 (Wiley-Interscience,
New York, 3rd ed., 1978) pp 277-297. Review of toxicology and human
exposure: Toxicological Profile for Acrolein (PB91-180307, 1990) 157 pp.
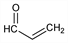
Properties
Liquid with pungent odor. Flammable, poisonous. mp 88°. d0 0.8621; d20 0.8389; d50 0.8075. bp760 52.5°; bp200 17.5°; bp100 2.5°; bp60 7.5°; bp1.0 64.5°. nD19 1.4022. Sol in 2 to 3 parts water; in alcohol, ether. Flash pt, open cup: <0°F (18°C). Vapor pressure at 20°: 210 mm Hg. Unstable, polymerizes
(especially under light or in the presence of alkali or strong acid)
forming disacryl, a plastic solid. Absorption spectrum: Lüthy, Z. Phys. Chem. 107, 291, 298 (1923).
LD50 orally in
rats: 0.046 g/kg (Smyth).Caution
Potential symptoms of overexposure
are irritation of eyes, skin and mucous membranes; decreased pulmonary
function; delayed pulmonary edema; chronic respiratory disease. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 6. See also Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins,
Baltimore, 5th ed., 1984) Section II, p 186.Use
Manuf colloidal forms of metals; making plastics,
perfumes; warning agent in methyl chloride refrigerant. Has been
used in military poison gas mixtures. Used in organic syntheses.
Aquatic herbicide.