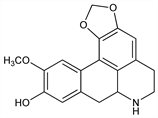
139. Actinodaphnine
Nomenclature
CAS number: 517-69-1
6,7,7a,8-Tetrahydro-11-methoxy-5H-benzo[g]-1,3-benzodioxolo[6,5,4-de]quinolin-10-ol; 10-methoxy-1,2-(methylenedioxy)-6aα-noraporphin-9-ol; 1,2-methylenedioxy-9-hydroxy-10-methoxynoraporphine. C18H17NO4; mol wt 311.33.
C 69.44%, H 5.50%, N 4.50%, O 20.56%.
Description and references
In bark of Actinodaphne hookeri Meissn., Lauraceae. Isoln: Krishna, Ghose, J. Indian Chem. Soc. 9, 429 (1932). Structure: Ghose et al., Helv. Chim. Acta 17, 919 (1934). Photolytic synthesis: M. S. Premila et al., Indian J. Chem. 13, 945 (1975).
Properties
Needles from alcohol, mp 211°. [α]D20 +33° (ethanol). Sol in acetone, alcohol, benzene, chloroform. Moderately sol in ether. Practically insol in water.Derivative
Hydrochloride.C18H17NO4.HCl; mol wt 347.79.
C 62.16%, H 5.22%, N 4.03%, O 18.40%, Cl 10.19%.
Properties
Needles from alcohol-ether, mp 281° (dec). [α]D20 +9°.Derivative
Methyl ether.C19H19NO4; mol wt 325.36.
C 70.14%, H 5.89%, N 4.30%, O 19.67%.