146. Acyclovir
Nomenclature
Description and references
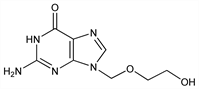
Orally active acyclic nucleoside with inhibitory activity towards several herpes viruses. Prepn: H. J. Schaeffer, DE 2539963; idem, US 4199574 (1976, 1980 to Wellcome). Convenient synthesis from guanine: H. Matsumoto et al., Chem. Pharm. Bull. 36, 1153 (1988) PubMed. Selectivity of action: G. B. Elion et al., Proc. Natl. Acad. Sci. USA 74, 5716 (1977) DOI PubMed. Chemistry, antiviral activity, metabolism: H. J. Schaeffer et al., Nature 272, 583 (1978) DOI PubMed. In vitro activity: P. Collins, D. J. Bauer, J. Antimicrob. Chemother. 5, 431 (1979) DOI PubMed. Effect on herpes simplex infections in mice: H. J. Field et al., Antimicrob. Agents Chemother. 15, 554 (1979) PubMed; on herpes zoster in immunocompromised patients: H. H. Balfour et al., N. Engl. J. Med. 308, 1448 (1983) PubMed. Treatment of primary episodes of genital herpes simplex infection: Y. J. Bryson et al., ibid. 916 PubMed; of recurrent genital herpes: S. E. Straus et al., ibid. 310, 1545 (1984) PubMed; J. M. Douglas et al., ibid. 1551 PubMed. HPLC determn in serum and clinical pharmacokinetics: G. Bahrami et al., J. Chromatogr. B 816, 327 (2005) DOI. Symposia on pharmacology and clinical studies: Am. J. Med. 73, Suppl. 1A, 1-392 (1982); J. Antimicrob. Chemother. 12, Suppl. B, 1-202 (1983); Scand. J. Infect. Dis. Suppl. 47, 1-176 (1985). Review: R. J. Whitley, J. W. Gnann, Jr., N. Engl. J. Med. 327, 782-789 (1992) PubMed.
Properties
Crystals from methanol, mp 256.5-257°. Maximum soly in water (37°): 2.5mg/ml. Sol in diluted hydrochloric acid; slightly sol in water. Insol in alcohol. pKa1 2.27. pKa2 9.25. LD50 in mice (mg/kg): >10,000 orally; 1000 i.p. (Schaeffer).Derivative
Sodium salt.Nomenclature
Properties
White crystalline powder. Maximum soly in water (25°): >100mg/ml.Therapeutic Category
Antiviral.
Therapeutic Category (Veterinary)
Antiviral.Keywords
Antiviral; Nucleosides/Nucleotides