151. Adefovir
Nomenclature
Description and references
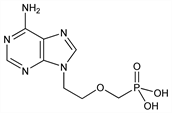
Acyclic nucleoside phosphonate (ANP) analog. Prepn: A. Holy, I. Rosenberg, EP 206459; eidem, US 4808716 (1986, 1989 both to Ceskoslov. Akad. Ved); eidem, Collect. Czech. Chem. Commun. 52, 2801 (1987); of dipivoxil ester prodrug: J. E. Starrett, Jr. et al., EP 481214 (1992 to Bristol-Myers Squibb); eidem, US 5663159 (1997 to Inst. Org. Chem. Biochem. Acad. Sci. Czech Rep.; Rega). Crystal structure: C. H. Schwalbe et al., J. Chem. Soc. Perkin Trans. 1 1991, 1348. HPLC determn: P. Augustijns et al., J. Liq. Chromatogr. Relat. Technol. 19, 2271 (1996). Clinical pharmacokinetics: K. C. Cundy et al., Antimicrob. Agents Chemother. 39, 2401 (1995). Review of pharmacology and therapeutic potential: L. Naesens et al., Antivir. Chem. Chemother. 8, 1-23 (1997). Clinical trial in HIV infected patients: J. Kahn et al., J. Am. Med. Assoc. 282, 2305 (1999); in hepatitis B virus (HBV): R. J. C. Gilson et al, J. Viral Hepat. 6, 387 (1999). Resistance surveillance in HBV infections: H. Yang et al., Hepatology 36, 464 (2002).
Properties
Off-white crystalline solid, mp >250°. pKa1 2.0, pKa2 6.8. uv max (H2O) 208, 260 nm (ε 19600, 14100); (0.1N HCl) 210, 260 nm (ε 19000, 13700); (0.1N NaOH) 216, 262 nm (ε 9600, 14500).Derivative
Di(pivaloyloxymethyl) ester.Nomenclature
Properties
Off-white crystalline powder. Soly in water (mg/ml): 19 (pH 2.0); 0.4 (pH 7.2). Log P (octanol/aq phosphate buffer): 1.91.Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides