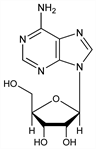
153. Adenosine
Nomenclature
Description and references
Nucleoside; widely distributed in nature. From yeast nucleic acid: Levene and Bass, Nucleic Acids (New York, 1931) p 163. Structure: Levene, Tipson, J. Biol. Chem. 94, 809 (1932); Bredereck, Ber. 66, 198 (1933); Z. Physiol. Chem. 223, 61 (1934); Gulland, Holiday, J. Chem. Soc. 1936, 765 DOI. See also: Szent-Gyrgyi, J. Physiol. 68, 213 (1930); Lythgoe et al., J. Chem. Soc. 1947, 355; 1948, 965 DOI DOI. Synthesis: Davoll et al., ibid. 1948, 967 DOI; H. Vorbrueggen, K. Krolikiewicz, Angew. Chem. Int. Ed. 14, 421 (1975) DOI. Crystal structure: T. F. Lai, R. E. Marsh, Acta Crystallogr. B28, 1982 (1972). Conformational properties: D. B. Davies, A. Rabczenko, J. Chem. Soc. Perkin Trans. 2 1975, 1703. Symposium on cardiac electrophysiology, pharmacology and clinical efficacy in supraventricular tachycardia: Prog. Clin. Biol. Res. 230, 1-395 (1987) PubMed. Reviews: see Adenine.
Properties
Crystals from water, mp 234-235°. [α]D11 61.7° (c = 0.706 in water); [α]9D 58.2° (c = 0.658 in water). uv max: 260 nm (ε 15100). Slightly sol in water. Practically insol in alcohol.Therapeutic Category
Antiarrhythmic.
Keywords
Antiarrhythmic