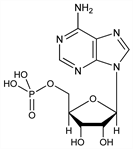
158. 5′-Adenylic Acid
Nomenclature
CAS number: 61-19-8
Muscle adenylic acid; ergadenylic acid; t-adenylic acid; adenosine 5′-monophosphate; adenosine phosphate; adenosine-5′-phosphoric acid; adenosine-5′-monophosphoric acid; A-5MP; AMP; NSC-20264; Adényl (Am. Home). C10H14N5O7P; mol wt 347.22.
C 34.59%, H 4.06%, N 20.17%, O 32.25%, P 8.92%.
Description and references
Nucleotide; widely distributed in nature. Prepn from tissues: Embden, Zimmerman, Z. Physiol. Chem. 167, 137 (1927); Embden, Schmidt, ibid. 181, 130 (1929); cf. Kalckar, J. Biol. Chem. 167, 445 (1947). Prepn by hydrolysis of ATP with barium hydroxide: Kerr, J. Biol. Chem. 139, 131 (1941). Synthesis: Baddiley, Todd, J. Chem. Soc. 1947, 648. Commercial prepn by enzymatic phosphorylation of adenosine. Monograph on synthesis of nucleotides: G. R. Pettit, Synthetic Nucleotides vol. 1 (Van Nostrand-Reinhold, New York, 1972) 252 pp. Crystal structure: Kraut, Jensen, Acta Crystallogr. 16, 79 (1963). Reviews: see Adenosine.
Properties
Crystals from water + acetone, mp 196-200°. [α]D20 47.5° (c = 2, 2% NaOH); [α]D20 26.0° (c = 2, 10% HCl). pK1 = 3.8; pK2 = 6.2. uv max (pH 7.0): 259 nm (aM 15.4 × 103). Readily sol in boiling water. The compound is readily deaminated by nitrous acid to form inosinic acid; less rapidly hydrolyzed than 3′-adenylic acid by sulfuric acid. Furfural is formed only in traces on distillation with 20% HCl, cf. Levene, Bass, Nucleic Acids (New York, 1931) pp 230-232.Therapeutic Category
Nutrient.