181. Aflatoxins G
Description and references
Toxic metabolites of fungi Aspergillus flavus Link ex Fries and Aspergillus parasiticus. Total synthesis of G1: Büchi, Weinreb, J. Am. Chem. Soc. 93, 746 (1971). Physical and chemical data: Hartley et al., Nature 198, 1056 (1963); Asao, J. Am. Chem. Soc. 87, 882 (1965). Toxicity data: R. B. A. Carnaghan et al., Nature 200, 1101 (1963). For general refs see Aflatoxins B.
Derivative
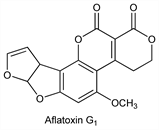
Aflatoxin G1.Nomenclature
CAS number: 1165-39-5
(7aR,10aS)-3,4,7a,10a-Tetrahydro-5-methoxy-1H,12H-furo[3′,2′:4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione. C17H12O7; mol wt 328.27.
C 62.20%, H 3.68%, O 34.12%.
Properties
Crystals, mp 244-246°. Exhibits green fluorescence. [α]D 556° (chloroform). uv max (ethanol): 243, 257, 264, 362 nm (ε 11500, 9900, 10000, 16100). LD50 orally in day old duckling: 39.2 μg/50 gm body wt (Carnaghan).Derivative
Aflatoxin G2.Nomenclature
CAS number: 7241-98-7
(7aR,10aS)-3,4,7a,9,10,10a-Hexahydro-5-methoxy-1H,12H-furo[3′,2′:4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione. C17H14O7; mol wt 330.29.
C 61.82%, H 4.27%, O 33.91%.
Description and references
The 9,10-dihydro deriv of aflatoxin G1.