194. Ajmaline
Nomenclature
Description and references
From roots of Rauwolfia serpentina (L.) Benth. (Ophioxylon serpentinum L.), Apocynaceae. Isolation: S. Siddiqui, R. H. Siddiqui, J. Indian Chem. Soc. 8, 667 (1931); 9, 539 (1932); 12, 37 (1935); L. van Itallie, A. J. Steenhauer, Arch. Pharm. 270, 313 (1932). Structure: A. Chatterjee, S. Bose, J. Indian Chem. Soc. 31, 17 (1954); F. A. L. Anet et al., J. Chem. Soc. 1954, 1242. Stereochemistry: M. F. Bartlett et al., J. Am. Chem. Soc. 84, 622 (1962). Synthesis: S. Masamune et al., ibid. 89, 2506 (1967); E. E. Van Tamelen, L. K. Oliver, ibid. 92, 2136 (1970); K. Mashimo, Y. Sato, Chem. Pharm. Bull. 18, 353 (1970). Physico-chemical properties: A. Petter, Arzneim.-Forsch. 24, 874 (1974). Antiarrhythmic activity: A. Petter, K. Engelmann, ibid. 876. Reviews: R. Robinson in Festschrift Arthur Stoll (Birkhuser-Verlag, Basel, 1957) pp 457-467; A. Koskinen, M. Lounasmaa in Progress in the Chemistry of Natural Products vol. 43, W. Herz et al., Eds. (Springer-Verlag, New York, 1983) pp 268-346.
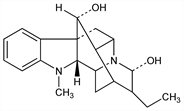
Properties
Pale amber, solvated, tetragonal prisms from methanol, C20H26N2O2.CH3OH, mp 158-160°. [α]D18 +131° (c = 0.4 in chloroform). Anhydr mp 205-207°. [α]D20 +144° (c = 0.8 in chloroform). uv max (ethanol): 247, 295 nm (log ε 3.94, 3.49). Sol in methanol, ethanol, ether, chloroform; slightly sol in water.Derivative
Hydrochloride dihydrate.Properties
Hexagonal bipyramidal crystals from water, mp 140°. [α]D18 +96.6°. One gram dissolves in 40 ml water.Therapeutic Category
Antihypertensive; antiarrhythmic.
Keywords
Antiarrhythmic; Antihypertensive