Nomenclature
CAS number: 107-95-9
β-Aminopropionic acid; 3-aminopropanoic acid; 3-aminopropionic acid; Abufène (Théraplix).
C
3H
7NO
2; mol wt 89.09.
C 40.44%, H 7.92%, N 15.72%, O 35.92%.
Description and references
Prepd by the action of KOBr and KOH upon succinimide:
Clarke, Behr, Org. Synth. 16, 1 (1936). By the action of liq ammonia upon methyl acrylate:
Morsch, Monatsh. Chem. 63, 220 (1933), cf. C.A. 41, 4104 (1947); by the addition of NH4OH
to acrylonitrile: Ford et al., J. Am. Chem. Soc. 69, 844 (1947). By electrolytic
oxidation of 3-amino-1-propanol in H2SO4 using
Pb electrodes without diaphragm: Jubilee
Vol. Emil Barell 1946, 85-91. For industrial
methods of prepn see several pats. by T. L. Gresham to
B. F. Goodrich. Prepn from ethylene cyanohydrin (β-hydroxypropionitrile):
Boatright, US 2734081 (1956 to Am. Cyanamid); from β-aminopropionitrile:
Ford, Org. Synth. coll.
vol. III, 34 (1955). Improved process: Beutel, Klemchuk, US 2956080 (1960 to Merck &
Co.).
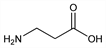
Properties
Orthorhombic bipyramidal crystals from water,
decomp 207° (very rapid heating). Decomp 197-198° (Ford, Org.
Syn. loc. cit.). Slightly sweet taste. pK1 3.60; pK2 10.19. pH of 5% aq soln: 6.0 to 7.3.
Freely sol in water, slightly in alcohol. Practically insol in ether,
acetone.Derivative
Hydrochloride.
C
3H
7NO
2.HCl; mol wt 125.55.
C 28.70%, H 6.42%, N 11.16%, O 25.49%, Cl 28.24%.
Properties
Plates, leaflets, mp 122.5°. Freely sol in water, less sol in
alcohol, insol in ether.Derivative
Platinichloride.
2C
3H
7NO
2.2HCl.PtCl
4; mol wt 588.00.
C 12.26%, H 2.74%, N 4.76%, O 10.88%, Cl 36.18%, Pt 33.18%.
Properties
Yellow leaflets from alcohol + HCl, decomp 210°.
Freely sol in water, sparingly in abs alcohol.Use
In the synthesis of pantothenic acid and derivatives;
as buffer in electroplating.
