216. Albuterol
Nomenclature
Description and references
β-Adrenergic agonist. Prepn: L. H. C. Lunts et al., ZA 6705591; eidem, US 3644353 (1968, 1972 both to Allen & Hanburys). Synthesis and structure-activity studies: D. T. Collin et al., J. Med. Chem. 13, 674 (1970) DOI PubMed. Prepn of isomers: C. J. Hawkins, G. T. Klease, ibid. 16, 856 (1973) DOI PubMed; R. P. Bakale, Spec. Chem. 15, 249 (1995). Absolute configuration of isomers: D. Hartley, D. Middlemiss, J. Med. Chem. 14, 895 (1971) DOI. Pharmacology: R. T. Brittain et al., Nature 219, 862 (1968) DOI PubMed; V. A. Cullum et al., Br. J. Pharmacol. 35, 141 (1969) PubMed. Metabolism: L. E. Martin et al., Eur. J. Pharmacol. 14, 183 (1971) DOI PubMed. Clinical comparison of levalbuterol with racemate: H. S. Nelson et al., J. Allergy Clin. Immunol. 102, 943 (1998) DOI PubMed. Comprehensive description: H. Y. Aboul-Enein et al., Anal. Profiles Drug Subs. 10, 665-689 (1981). Review of clinical experience in asthma and in preterm labor: A. H. Price, S. P. Clissold, Drugs 38, 77-122 (1989) DOI PubMed; of toxicology: S. E. Libretto, Arch. Toxicol. 68, 213-216 (1994) DOI PubMed. Review of clinical trials for add-on therapy to inhaled steroids: S. Shrewsbury et al., Br. Med. J. 320, 1368-1373 (2000) DOI.
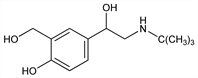
Properties
Crystalline powder from ethanol-ethyl acetate or ethyl acetate-cyclohexane, mp 151° (Lunts); 157-158° (Collin). Sol in alcohol, most organic solvents. Sparingly sol in water.Derivative
Sulfate.Nomenclature
Properties
White crystalline powder. Sol in water; slightly sol in ethanol.Derivative
(R)-Form.Nomenclature
Derivative
(R)-Form hydrochloride.Nomenclature
Properties
Crystallized as the monohydrate; changes crystalline form at 175°; dec 188-195°. [α]D20 32.2° (c = 0.1 in water).Therapeutic Category
Bronchodilator; tocolytic.
Therapeutic Category (Veterinary)
Bronchodilator.Keywords
β-Adrenergic Agonist; Bronchodilator; Ephedrine Derivatives; Tocolytic