220. Alclometasone
Nomenclature
CAS number: 67452-97-5
(7α,11β,16α)-7-Chloro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; 7α-chloro-16α-methylprednisolone. C22H29ClO5; mol wt 408.92.
C 64.62%, H 7.15%, Cl 8.67%, O 19.56%.
Description and references
Non-fluorinated corticosteroid with low systemic effects. Prepn: M. J. Green et al., US 4076708; M. J. Green, H. J. Shue, US 4124707 (both 1978 to Schering); M. J. Green et al., J. Steroid Biochem. 11, 61 (1979); H. J. Shue et al., J. Med. Chem. 23, 430 (1980). Topical anti-inflammatory activity of the 17,21-dipropionate: B. Lutsky et al., Arzneim.-Forsch. 29, 992 (1979); M. J. Green et al., ibid. 30, 1618 (1980).
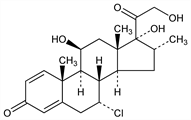
Properties
Crystals from acetone/hexane, mp 176-179°. [α]D26 +47.5° (c = 0.3 in DMF). uv max (methanol): 242 nm (ε 15500).Derivative
17,21-Dipropionate.Nomenclature
CAS number: 66734-13-2
Sch-22219; Aclosone (Schering); Aclovate (GSK); Almeta (Shionogi); Delonal (Essex); Legederm (Schering); Modrasone (Schering); Perderm (Schering). C28H37ClO7; mol wt 521.04.
C 64.54%, H 7.16%, Cl 6.80%, O 21.49%.
Properties
Crystals from acetone/methanol/isopropyl ether, mp 212-216°. [α]D26 +42.6° (c = 0.3 in DMF). uv max (methanol): 242 nm (ε 15600).Therapeutic Category
Anti-inflammatory (topical).
Keywords
Glucocorticoid