231. Aleuritic Acid
Nomenclature
CAS number: 533-87-9
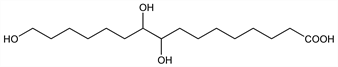
(9R,10S)-rel-9,10,16-Trihydroxyhexadecanoic
acid; 9,10,16-trihydroxypalmitic acid; 8,9,15-trihydroxypentadecane-1-carboxylic acid. C16H32O5; mol wt 304.42.
C 63.13%, H 10.60%, O 26.28%.
Description and references
One of the constituent acids of shellac, q.v. Obtained in 43% yield from dewaxed shellac: Schaeffer, Gardner, Ind. Eng. Chem. 30, 333 (1938); Gidvani, J. Chem. Soc. 1944, 306; Sengupta, Bose, J. Sci. Ind. Res. 11B, 458 (1952). The acid obtained from shellac is optically inactive, although it contains two asymmetric carbon atoms. It has been shown to be the dl-erythro or dl-cis form, and is the only form described here. Synthesis of diastereoisomers: Mitter et al., Sci. Cult. 8, 273 (1942); Hunsdiecker, Ber. 76, 142 (1943); 77, 185 (1944); Baudart, Compt. Rend. 221, 205 (1945).
Properties
Crystals from dilute ethanol, mp 100-101°. Sol in methanol. Forms a crystalline sodium salt.Derivative
Methyl ester.C17H34O5; mol wt 318.45.
C 64.12%, H 10.76%, O 25.12%.
Properties
Fine feathery needles, mp 72-73°; bp0.1 235°. Sol in methanol, ethanol, chloroform, acetone. Less sol in benzene. Insol in petr ether.Derivative
Ethyl ester.Nomenclature
CAS number: 6003-09-4
C18H36O5; mol wt 332.48.
C 65.02%, H 10.91%, O 24.06%.
Properties
Needles from dil ethanol, mp 59°.Derivative
Hydrazide.Nomenclature
CAS number: 6003-10-7
C16H34N2O4; mol wt 318.45.
C 60.35%, H 10.76%, N 8.80%, O 20.10%.