238. Alfuzosin
Nomenclature
Description and references
α1-Adrenoceptor antagonist structurally similar to prazosin, q.v. Prepn: P. M. J. Manoury, DE 2904445; idem, US 4315007 (1979, 1982 both to Synthelabo); and antihypertensive activity in rats: P. M. Manoury et al., J. Med. Chem. 29, 19 (1986). Pharmacology: A. G. Ramage, Eur. J. Pharmacol. 129, 307 (1986). HPLC determn in biological fluids: P. Guinebault et al., J. Chromatogr. 353, 361 (1986). Pharmacology in humans: A. H. Deering, Br. J. Clin. Pharmacol. 25, 417 (1988). Clinical evaluation in essential hypertension: S. Leto Di Priolo et al., Eur. J. Clin. Pharmacol. 35, 25 (1988); A. K. Ghosh, S. Ghosh, Ger. Cardiovasc. Med. 1, 81 (1988). Clinical trial in benign prostatic hyperplasia (BPH): C. G. Roehrborn et al., BJU Int. 92, 257 (2003). Review of clinical experience in BPH: D. M. Weiner, F. C. Lowe, Expert Opin. Pharmacother. 4, 2057-2063 (2003).
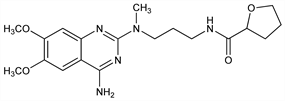
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol + ether, mp 225° (Manoury, 1986), also reported earlier as mp 235° (dec) (Manoury, 1982). pKa 8.13. Slightly hygroscopic. Freely sol in water; sparingly sol in alcohol. Practically insol in methylene chloride.Therapeutic Category
Antihypertensive. In treatment of benign prostatic hypertrophy.
Keywords
Antihypertensive; Quinazoline Derivatives; Antiprostatic Hypertrophy; α-Adrenergic Blocker