239. Algestone
Nomenclature
CAS number: 595-77-7
(16α)-16,17-Dihydroxypregn-4-ene-3,20-dione; 16α,17-dihydroxyprogesterone; 4-pregnene-16α,17α-diol-3,20-dione; alphasone. C21H30O4; mol wt 346.46.
C 72.80%, H 8.73%, O 18.47%.
Description and references
Prepn from 16-dehydroprogesterone: Inhoffen et al., Ber. 87, 593 (1954); Cooley et al., J. Chem. Soc. 1955, 4373; Allen, Bernstein, J. Am. Chem. Soc. 78, 1909 (1956); Hydorn et al., Steroids 3, 493 (1964). Manuf from 16-dehydroprogesterone: Colton, US 2727909 (1955 to Searle); Hydorn et al., US 3165541 (1965 to Olin Mathieson); from its 16α-acetate: Diassi, US 3027384 (1962 to Olin Mathieson).
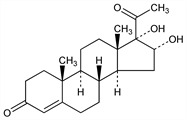
Properties
Needles from ethanol + dichloromethane, mp 225°. [α]D22 +95° (c = 0.81 in CHCl3). uv max: 240 nm (ε 16600).Derivative
Cyclic acetal with acetone.Nomenclature
CAS number: 4968-09-6
16α,17-[(1-Methylethylidene)bis(oxy)]pregn-4-ene-3,20-dione; algestone acetonide; alphasone acetonide; 16α,17α-isopropylidenedioxyprogesterone. C24H34O4; mol wt 386.52.
C 74.58%, H 8.87%, O 16.56%.
Description and references
Prepn: Cooley et al., loc. cit; Fried et al., Chem. Ind. (London) 1961, 465.
Properties
Needles from aq ethanol, mp 210°. [α]D20 +137° (c = 0.7 in CHCl3).Derivative
16α-Methyl ether.Nomenclature
17-Hydroxy-16α-methoxypregn-4-ene-3,20-dione.C22H32O4; mol wt 360.49.
C 73.30%, H 8.95%, O 17.75%.
Description and references
Prepd from the free diol via 16α,17-dihydroxypregn-4-ene-3,20-dione cyclic borate: Fried, US 3006930 (1961 to Olin Mathieson).
Properties
Crystals from 95% ethanol, mp 142-143°. [α]D23 +60° (c = 0.15 in CHCl3). uv max: 234 nm (ε 15400).Therapeutic Category
Acetonide as anti-inflammatory (topical).
Keywords
Glucocorticoid