251. Alizarin
Nomenclature
CAS number: 72-48-0
1,2-Dihydroxy-9,10-anthracenedione; 1,2-dihydroxyanthraquinone; C.I. Mordant Red 11; C.I. Pigment Red 83; C.I. 58000. C14H8O4; mol wt 240.21.
C 70.00%, H 3.36%, O 26.64%.
Description and references
Occurs in the root of the madder plant (Rubia tinctorum L., Rubiaceae; Krappwurzel) in combination with 2 mols glucose, called ruberythric acid. Was known and used in ancient Egypt, Persia, and India. Synthesized from 2-anthraquinonesulfonic acid sodium salt : Caro et al., Ber. 3, 359 (1870); Perkin, Ber. 9, 281 (1876). Historical review: Fieser, J. Chem. Educ. 7, 2609 (1930). Laboratory prepn: Gattermann-Wieland, Laboratory Methods of Organic Chemistry (New York, 1937). Modern methods of manufacture: Pohl, Ullmanns Encyklopdie der technischen Chemie vol. I, p 200; Fierz-David and Blangey, Grundlegende Operationen der Farbenchemie (Vienna, 5th ed., 1943). See also Colour Index vol. 4, (3rd ed., 1971) p 4513.
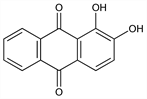
Properties
Orthorhombic, orange needles by sublimation or from abs alc. Solvated scales from dil alc or by evaporation from ether. Sublimes at 110° (2 mm Hg). mp 290°. bp 430°. Absorption spectrum: Moir, J. Chem. Soc. 1927, 1810. Soly in water at 18°: 2.1×106 mols/l; at 25°: 2.5×106 mols/l. Sol in 300 parts boiling water; moderately sol in alcohol, freely in hot methanol and in ether at 25°. Also sol in benzene, toluene, xylene, pyridine, carbon disulfide, glacial acetic acid. Sol in water solns of alkalies with blue color, but without fluorescence. Fluorescent solns indicate unchanged 2-anthraquinone sodium sulfonate.Derivative
1-Methyl ether.C15H10O4; mol wt 254.24.
C 70.86%, H 3.96%, O 25.17%.
Properties
Orange needles with 1H2O from dil methanol. When dried at 100° mp 179°.Derivative
2-Methyl ether.C15H10O4; mol wt 254.24.
C 70.86%, H 3.96%, O 25.17%.
Properties
Orange needles from alcohol, mp 231°.Derivative
Dimethyl ether.C16H12O4; mol wt 268.26.
C 71.64%, H 4.51%, O 23.86%.