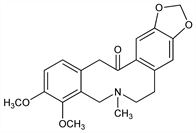
265. Allocryptopine
Nomenclature
CAS number: 485-91-6
5,7,8,15-Tetrahydro-3,4-dimethoxy-6-methyl[1,3]benzodioxolo[5,6-e][2]benzazecin-14(6H)-one; 5,7,8,15-tetrahydro-3,4-dimethoxy-6-methylbenzo[e][1,3]dioxolo[4,5-k][3]benzazecin-14(6H)-one. C21H23NO5; mol wt 369.41.
C 68.28%, H 6.28%, N 3.79%, O 21.66%.
Description and references
Isomeric with cryptopine, q.v. Obtained from Chelidonium majus L., Bocconia cordata Willd., Sanguinaria canadensis L. and allied Papaveraceae: Manske in Manske, Holmes, The Alkaloids vol. IV (Academic Press, New York, 1954) p 159. Structure: Gadamer, Arch. Pharm. 257, 298 (1919). Exists in two isomeric modifications designated as α- and β-forms. Identity of α-form with α-fagarine: Redemann et al., J. Am. Chem. Soc. 71, 1030 (1949). Synthesis: Haworth, Perkin, J. Chem. Soc. 1926, 445; Bentley, Murray, ibid. 1963, 2497.