858. Atazanavir
Nomenclature
Description and references
Azapeptide HIV protease inhibitor. Prepn: A. Fssler et al., WO 9740029; eidem, US 5849911 (1997, 1998 both to Novartis); G. Bold et al., J. Med. Chem. 41, 3387 (1998); of bisulfate salt: J. Singh et al., WO 9936404; eidem, US 6087383 (1999, 2000 both to Bristol-Myers Squibb); Z. Xu et al., Org. Process Res. Dev. 6, 323 (2002). Comparative anti-HIV activity: B. S. Robinson et al., Antimicrob. Agents Chemother. 44, 2093 (2000). HPLC determn in plasma: E. Cateau et al., J. Pharm. Biomed. Anal. 39, 791 (2005). Clinical evaluation in HIV: I. Sanne et al., J. Acquir. Immune Defic. Syndr. 32, 18 (2003). Review of pharmacology and clinical efficacy in HIV: C. Le Tiec et al., Clin. Pharmacokinet. 44, 1035-1050 (2005); T. S. Harrison, L. J. Scott, Drugs 65, 2309-2336 (2005).
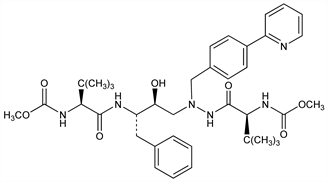
Properties
Crystals from ethanol/water, mp 207-209°. [α]D 47° (c = 1 in ethanol). Insol in water (<1 μg/ml at 24°).Derivative
Sulfate.Nomenclature
Properties
Crystals from ethanol + heptane, mp 195.0°, or acetone, mp 198-199° (dec). [α]D22 46.1° (c = 1 in 1:1 CH3OH/H2O, pH = 2.6). Soly in water: 4-5 mg/ml.Therapeutic Category
Antiretroviral.
Keywords
Antiretroviral; Protease Inhibitors; HIV Protease Inhibitor