310. Alphaprodine
Nomenclature
CAS number: 77-20-3
(cis)-1,3-Dimethyl-4-phenyl-4-piperidinol propanoate; (±)-α-1,3-dimethyl-4-phenyl-4-piperidinyl propionate; dl-α-1,3-dimethyl-4-phenyl-4-propionoxypiperidine; (±)-α-prodine. C16H23NO2; mol wt 261.36.
C 73.53%, H 8.87%, N 5.36%, O 12.24%.
Description and references
Mixture of the two cis isomers of prodine; betaprodine is the mixture of trans isomers. Prepn: J. Lee, A. Ziering, US 2498433 (1950 to Hoffmann-La Roche); A. H. Beckett et al., J. Pharm. Pharmacol. 9, 939 (1957); A. Ziering et al., J. Org. Chem. 22, 1521 (1957). Configurational studies: F. R. Ahmed et al., Chem. Ind. (London) 1959, 485; eidem, ibid. 1962, 97. Stereostructure-activity studies: M. M. Abdel-Monem et al., J. Med. Chem. 15, 494 (1972). Pharmacology and toxicology: G. M. Gruber et al., J. Pharmacol. Exp. Ther. 99, 312 (1950). Review of clinical experience: R. C. Lunt, H. E. Howard, Pediatr. Dent. 10, 121-126 (1988).
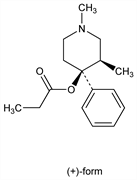
Derivative
Hydrochloride.Nomenclature
CAS number: 561-78-4
Nu-1196; Nisentil (Roche). C16H23NO2.HCl; mol wt 297.82.
C 64.53%, H 8.12%, N 4.70%, O 10.74%, Cl 11.90%.
Properties
Crystals from acetone, mp 220-221°. Slightly saline taste. Freely sol in water, alc, chloroform. Practically insol in ether. pH of 1% aq soln 4.5-5.2. LD50 in mice: 54 mg/kg i.v., 73 mg/kg i.p.; in rats: 22 mg/kg i.p. (Gruber).Derivative
Betaprodine hydrochloride.Nomenclature
CAS number: 49638-23-5
(±)-β-Prodine hydrochloride; Nu-1779. Properties
Crystals from methyl ethyl ketone, mp 195-196° (Beckett); from acetone + methanol, mp 199-200° (Ziering).Note
These are controlled substances (opiates): 21 CFR, 1308.11 (betaprodine) and 1308.12 (alphaprodine).Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Phenylpiperidines