320. Altretamine
Nomenclature
Description and references
An antitumor agent which also acts as a chemosterilant for male houseflies and other insects. Synthesis: A. W. Hofmann, Ber. 18, 2755 (1885); D. W. Kaiser et al., J. Am. Chem. Soc. 73, 2984 (1951); Y. Bessiere-Chretien, H. Serne, Bull. Soc. Chim. Fr. 6, 2039 (1973). Production process: H. von Brachel, H. Kindler, DE 1240870; eidem, US 3424752 (1967, 1969 both to Casella Farbwerke). Chemosterilant effect: S. C. Chang et al., Science 144, 57 (1964); A. De Milo et al., J. Econ. Entomol. 65, 1548 (1972). Chromatographic detn in plasma or serum: A. Hulshoff et al., J. Chromatogr. 181, 363 (1980). Disposition and metabolism in rabbits and humans: M. M. Ames et al., Cancer Res. 39, 5016 (1979). Evaluation in breast cancer: C. J. Fabian et al., Cancer Treat. Rep. 63, 1359 (1979); in ovarian cancer: J. T. Wharton et al., Am. J. Obstet. Gynecol. 133, 833 (1979). Mammalian toxicity study: R. L. Jasper et al., Fed. Proc. 24, 641 (1965). Toxicological studies in dogs and mice: D. C. Thake et al., Gov. Rep. Announce. Index (USA) 79, 92 (1979). In vitro cytotoxicity study: M. D'Incalci et al., Br. J. Cancer 41, 630 (1980).
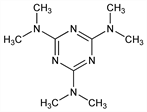
Properties
Needles from abs ethanol, mp 172-174°. uv max (ethanol): 226 nm (ε 49400), H. J. Anderson et al., Can. J. Chem. 49, 2315 (1971). Sol in chloroform. Insol in water. LD50 in rats, guinea pigs (mg/kg): 350, 255 orally (Jasper).Use
As exptl insect chemosterilant.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkylating Agents; Ethylenimines and Methylmelamines