390. Amdinocillin Pivoxil
Nomenclature
CAS number: 32886-97-8
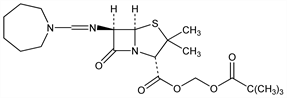
(2S,5R,6R)-6-[[(Hexahydro-1H-azepin-1-yl)methylene]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid (2,2-dimethyl-1-oxopropoxy)methyl ester; pivaloyloxymethyl 6-[(hexahydro-1H-azepin-1-yl)methyleneamino]penicillanate; pivamdinocillin; pivmecillinam; FL-1039; Selexid (susp.) (Leo Pharm). C21H33N3O5S; mol wt 439.57.
C 57.38%, H 7.57%, N 9.56%, O 18.20%, S 7.29%.
Description and references
Semi-synthetic antibiotic related to penicillin. Pivaloyloxymethyl ester of amdinocillin, q.v. Prepn: F. J. Lund, DE 2055531; idem, US 3957764 (1971, 1976 both to Lvens Kemiske Fabrik). HPLC stability analysis: R. B. Hagel, E. H. Waysek, J. Chromatogr. 178, 97 (1979). Bacteriological and pharmacokinetic study: T. Damsgaard et al., J. Antimicrob. Chemother. 5, 267 (1979). Metabolism: J. D. Anderson, M. A. Adams, Chemotherapy 25, 1 (1979). Clinical study: B. T. Andersen et al., Infection 8, 27 (1980). Toxicity study: S. Sato et al., Takeda Kenkyushoho 35, 179 (1976), C.A. 86, 115154w (1977).
Properties
Crystals from cyclohexane, mp 118.5-119.5°. [α]D20 +231° (c = 1 in 96% ethanol). LD50 in mice, rats (mg/kg): 475-480, 465 i.v.; 1736-1930, 1935-2100 s.c.; 3020, 9500-10000 orally (Sato).Derivative
Hydrochloride.Nomenclature
CAS number: 32887-03-9
Selexid (tabl.) (Leo Pharm); Melysin (Takeda). C21H33N3O5S.HCl; mol wt 476.03.
C 52.99%, H 7.20%, N 8.83%, O 16.81%, S 6.74%, Cl 7.45%.
Properties
Crystals from methanol-diisopropyl ether, mp 172-173°. [α]D20 +219° (c = 1 in 0.1N HCl).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins