399. Amidephrine
Nomenclature
CAS number: 37571-84-9
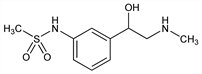
N-[3-[1-Hydroxy-2-(methylamino)ethyl]phenyl]methanesulfonamide; 3′-[1-hydroxy-2-(methylamino)ethyl]methanesulfonanilide; MJ-1996. C10H16N2O3S; mol wt 244.31.
C 49.16%, H 6.60%, N 11.47%, O 19.65%, S 13.12%.
Description and references
α1 Adrenoceptor agonist. Prepn: Larsen, Uloth, FR M3027 (1965 to Mead Johnson), C.A. 62, 13091a (1965); Uloth et al., J. Med. Chem. 9, 88 (1966) DOI PubMed. Pharmacology of the racemate: Dungan et al., Int. J. Neuropharmacol. 4, 219 (1965) DOI PubMed; Stanton et al., ibid. 235 DOI PubMed; of the isomers: Larsen, Lish, Nature 203, 1283 (1964) DOI PubMed; Buchthal, Jenkinson, Eur. J. Pharmacol. 10, 293 (1970) DOI PubMed. Toxicology: J. H. Weikel, Jr., K. H. Harper, Toxicol. Appl. Pharmacol. 23, 589 (1972) DOI PubMed.
Properties
Crystals, mp 159-161°. pKa 9.1.Derivative
Methanesulfonate.Nomenclature
CAS number: 1421-68-7
Amidephrine mesylate; MJ-5190; Fentrinol (Frika). C10H16N2O3S.CH3SO3H; mol wt 340.42.
C 38.81%, H 5.92%, N 8.23%, O 28.20%, S 18.84%.
Properties
Crystals from ethanol, mp 207-209°. LD50 in mice (mg/kg): 2284 orally, 780 i.p., 190 i.v., 1990 s.c.; in male, female Mead Johnson rats (mg/kg): 229, 36 orally; 144, 25 i.p.; in male, female Sprague-Dawley rats (mg/kg): 24, 13 orally; in rabbits (mg/kg): 12 orally, 7.5 intradermally (Weikel, Harper).Therapeutic Category
Vasoconstrictor; decongestant (nasal).
Keywords
α-Adrenergic Agonist; Decongestant