405. Amikacin
Nomenclature
Description and references
Semisynthetic aminoglycoside antibiotic derived from kanamycin A. Prepn: Kawaguchi et al., J. Antibiot. 25, 695 (1972); H. Kawaguchi, T. Naito, DE 2234315; H. Kawaguchi et al., US 3781268 (both 1973 to Bristol-Myers). Biological formation from kanamycin A: L. M. Cappelletti, R. Spagnoli, J. Antibiot. 36, 328 (1983). Microbiological evaluation: Price et al., ibid. 25, 709 (1972). Pharmacokinetics: Cabana, Taggart, Antimicrob. Agents Chemother. 3, 478 (1973). In vitro studies: Yu, Washington, ibid. 4, 133 (1973); Bodey, Stewart, ibid. 186. Pharmacology in humans: Bodey et al., ibid. 5, 508 (1974). Toxicity studies: Fujisawa et al., J. Antibiot. 27, 677 (1974). Review: K. A. Kerridge in Pharmacological and Biochemical Properties of Drug Substances vol. 1, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1977) pp 125-153. Comprehensive description: P. M. Monteleone et al., Anal. Profiles Drug Subs. 12, 37-71 (1983).
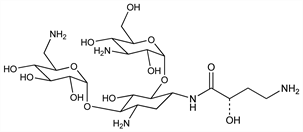
Properties
White crystalline powder from methanol-isopropanol, mp 203-204° (sesquihydrate). [α]D23 +99° (c = 1.0 in water). LD50 in mice of solns pH 6.6, pH 7.4 (mg/kg): 340, 560 i.v. (Kawaguchi).Derivative
Sulfate.Nomenclature
Properties
Amorphous form, dec 220-230°. [α]D22 +74.75° (water).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Aminoglycosides