407. Aminacrine
Nomenclature
CAS number: 90-45-9
9-Acridinamine; 5-aminoacridine; 9-aminoacridine. C13H10N2; mol wt 194.23.
C 80.39%, H 5.19%, N 14.42%.
Description and references
Prepn: A. Albert, B. Ritchie, Org. Synth. coll. vol. III, 53 (1955). Toxicity study: D. C. Brodie, E. Lowenhaupt, J. Am. Pharm. Assoc. 38, 498 (1949). Spectrophotometric determn in pharmaceuticals: E. A. Bunch, J. Assoc. Off. Anal. Chem. 66, 140 (1983). DNA intercalator used to induce exptl frameshift mutations: M. Conrad, M. D. Topal, J. Biol. Chem. 261, 16226 (1986).
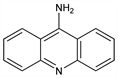
Properties
Sulfur-yellow needles from alcohol or acetone, mp 241°. Moderately strong base. pKa (25°): 4.53. Freely sol in alcohol; slightly sol in chloroform, toluene, pyridine; sol in acetone.Derivative
Hydrochloride.Nomenclature
CAS number: 134-50-9
Monacrin (Sterling Winthrop). C13H10N2.HCl; mol wt 230.69.
C 67.68%, H 4.81%, N 12.14%, Cl 15.37%.
Properties
Pale yellow crystals. Neutral reaction. One of the most highly fluorescent substances. One gram dissolves in 300 ml water giving a faintly yellow soln showing bluish-violet fluorescence. LD50 orally in mice: 78 mg/kg (Brodie, Lowenhaupt).Therapeutic Category
Antiseptic.
Therapeutic Category (Veterinary)
Antiseptic.Keywords
Antiseptic/Disinfectant; Dyes