453. 4-Amino-1-naphthol
Nomenclature
CAS number: 2834-90-4
4-Amino-1-naphthalenol; 4-hydroxy-α-naphthylamine. C10H9NO; mol wt 159.18.
C 75.45%, H 5.70%, N 8.80%, O 10.05%.
Description and references
Prepd by treating α-naphthol with benzenediazonium chloride and reducing the benzeneazo-α-naphthol with sodium hydrosulfite: Conant et al., Org. Synth. 3, 7 (1923); cf. Fieser, Fieser, J. Am. Chem. Soc. 57, 493 (1935). From α-naphthylhydroxylamine by rearrangement in acetone: Neunhoffer, Liebich, Ber. 71B, 2247 (1938).
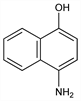
Properties
Needles. Unless kept absolutely dry, it acquires a violet discoloration on storage and oxidizes to 1,4-naphthoquinone. Usually isolated as the hydrochloride, C10H9NO.HCl, needles, very sol in water.Derivative
N-Acetyl deriv.Nomenclature
4-Acetamido-1-naphthol; naphthacetol.C12H11NO2; mol wt 201.22.
C 71.63%, H 5.51%, N 6.96%, O 15.90%.