572. Amodiaquin
Nomenclature
CAS number: 86-42-0
4-[(7-Chloro-4-quinolinyl)amino]-2-[(diethylamino)methyl]phenol; 4-[(7-chloro-4-quinolyl)amino]-α-(diethylamino)-o-cresol; 7-chloro-4-(3-diethylaminomethyl-4-hydroxyanilino)quinoline; 7-chloro-4-(3-diethylaminomethyl-4-hydroxyphenylamino)quinoline; 4-(3′-diethylaminomethyl-4′-hydroxyanilino)-7-chloroquinoline; SN-10751. C20H22ClN3O; mol wt 355.86.
C 67.50%, H 6.23%, Cl 9.96%, N 11.81%, O 4.50%.
Description and references
Prepd from 4,7-dichloroquinoline and 4-acetamido-α-diethylamino-o-cresol: Burckhalter et al., J. Am. Chem. Soc. 70, 1363 (1948); US 2474819; US 2474821 (1949 to Parke, Davis). Alternate synthesis from 2-aminomethyl-p-aminophenol and 4,7-dichloroquinoline: Natarajan, Lan, Arzneim.-Forsch. 22, 1230 (1972). Spectral study: J. Kracmar et al, Pharmazie 29, 773 (1974). HPLC determn in plasma, blood and urine: E. Pussard et al., J. Chromatogr. 374, 111 (1986). Comprehensive description: I. Ahmad et al., Anal. Profiles Drug Subs. 21, 43-73 (1992).
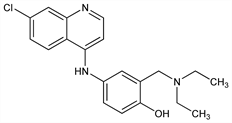
Properties
Crystals from abs. ethanol, mp 208° (dec).Derivative
Dihydrochloride dihydrate.Nomenclature
CAS number: 6398-98-7
CAM-AQ1; Camoquin (Parke-Davis); Flavoquine (Aventis). C20H22ClN3O.2HCl.2H2O; mol wt 464.81.
C 51.68%, H 6.07%, Cl 22.88%, N 9.04%, O 10.33%.
Properties
Yellow, bitter crystals, dec 150-160°. uv max (methanol): 342 nm (E1%1cm 349); (water): 341.5 nm (E1%1cm 389); (0.1N HCl): 342 nm (E1%1cm 396). Sol in water; sparingly sol in alc; very slightly sol in benzene, chloroform, ether. pH of 1% aq soln 4.0 to 4.8.Derivative
Dihydrochloride hemihydrate.Properties
Yellow crystals from methanol, mp 243°. Slightly sol in water, alc.Therapeutic Category
Antimalarial.
Keywords
Antimalarial