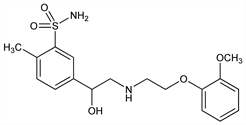
575. Amosulalol
Nomenclature
Description and references
Sulfonamide-substituted phenylethanolamine with α- and β-adrenergic blocking activity. Prepn: K. Imai et al., DE 2843016; eidem, US 4217305 (1979, 1980 both to Yamanouchi). Pharmacology and receptor blocking activity: T. Takenaka et al., Eur. J. Pharmacol. 85, 35 (1982); of isomers: K. Honda et al., J. Pharmacol. Exp. Ther. 236, 776 (1986). GC determn in urine: H. Kamimura et al., J. Chromatogr. 275, 81 (1983); HPLC determn in plasma: H. S. Gwak et al., J. Chromatogr. B 818, 109 (2005). Clinical pharmacokinetics: M. Nakashima et al., Clin. Pharmacol. Ther. 36, 436 (1984); metabolism: H. Kamimura et al., Xenobiotica 15, 413 (1985). Clinical evaluation in hypertension: K. Ando et al., J. Cardiovasc. Pharmacol. 20, 7 (1992).
Derivative
Monohydrochloride.Nomenclature
Properties
Colorless crystals, mp 158-160°. pK1 7.4; pK2 10.2.Derivative
R-()-Form hydrochloride.Properties
mp 158°. [α]D20 30.4° (c = 1 in methanol).Derivative
S-(+)-Form hydrochloride.Properties
mp 158°. [α]D20 +30.7° (c = 1 in methanol).Therapeutic Category
Antihypertensive.
Keywords
α-Adrenergic Blocker; β-Adrenergic Blocker; Antihypertensive; Arylethanolamine Derivatives