594. Amsacrine
Nomenclature
Description and references
Cytostatic agent with antiviral and immunosuppressive properties. Prepn: B. F. Cain et al., J. Med. Chem. 18, 1110 (1975). Synthesis and biological properties of spin-labeled amsacrine: B. K. Sinha et al., ibid. 19, 994 (1976). Mechanism of action: W. R. Wilson, Chem. N. Z. 37, 148 (1973). Pharmacologic disposition: R. L. Cysyk et al., Drug Metab. Dispos. 5, 579 (1977). Antiviral activity: D. M. Byrd, Ann. N.Y. Acad. Sci. 284, 463 (1977). Immunosuppressive properties: B. C. Baguley et al., Eur. J. Cancer 10, 169 (1974). Exptl antitumor properties: B. F. Cain, G. J. Atwell, ibid. 539. Efficacy in adult acute leukemia: S. S. Legha et al., Ann. Intern. Med. 93, 17 (1980). Toxicologic studies: K. L. Pavkov et al., U.S. NTIS Report PB-298106 (1979) 284 pp, C.A. 92, 34335c (1980). Review of pharmacology and clinical efficacy: J. Hornedo, D. A. Van Echo, Pharmacotherapy 5, 78-90 (1985).
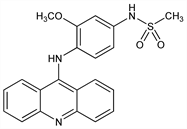
Properties
LD50 in male, female CDF1 mice: 810 mg/m2; 729 mg/m2 orally (Pavkov).Derivative
Hydrochloride.Nomenclature
NSC-141549.Properties
Crystals, mp 197-199°. LD50 i.p. in mice: ≈60 mg/kg (Byrd).Derivative
Methanesulfonate.Nomenclature
NSC-156303.Properties
Crystals, mp 292-293°. LD50 i.p. in mice: ≈24 mg/kg (Byrd).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Topoisomerase II Inhibitor