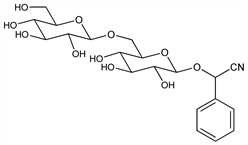
597. Amygdalin
Nomenclature
Description and references
The name amygdalin is currently used interchangeably with laetrile. Cyanogenic glycoside which occurs in seeds of Rosaceae; principally in bitter almonds; also in peaches and apricots. Most common constituent of Laetrile preparations. Structure and synthesis: W. N. Haworth, B. Wylam, J. Chem. Soc. 123, 3120 (1923); Kuhn, Ber. 56, 857 (1923); R. Campbell, W. N. Haworth, J. Chem. Soc. 125, 1337 (1924); Hudson, J. Am. Chem. Soc. 46, 483 (1924); Zemplén, Kunz, Ber. 57, 1357 (1924); Kuhn, Sobotka, ibid. 1767; Baumann, Pigman, The Carbohydrates, W. Pigman, Ed. (Academic Press, New York, 1957) p 550. Enzymic hydrolysis studies: Haisman, Knight, Biochem. J. 103, 528 (1967). The term Laetrile has also been applied to mandelonitrile β-glucuronide. Purported prepn: E. T. Krebs, E. T. Krebs, Jr., GB 788855 (1958) and US 2985664 (1961). Synthesis, characterization and comparison of mandelonitrile β-glucuronide with amygdalin: C. Fenselau et al., Science 198, 625 (1977). Pharmacology and cyanide toxicity studies of amygdalin (laetrile): C. G. Moertel et al., J. Am. Med. Assoc. 245, 591 (1981); M. M. Ames et al., Cancer Chemother. Pharmacol. 6, 51 (1981). Pharmacokinetics: A. G. Rauws et al., Arch. Toxicol. 49, 311 (1982). Determn methods in tissues and fluids: J. Balkon, J. Anal. Toxicol. 6, 244 (1982). Amygdalin (laetrile) is a toxic drug that is not effective as a cancer treatment: C. G. Moertel et al., N. Engl. J. Med. 306, 201 (1982). Review of the controversial use of amygdalin (laetrile): V. Herbert, Am. J. Clin. Nutr. 32, 1121-1158 (1979).