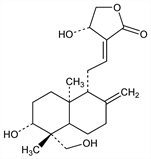
633. Andrographolide
Nomenclature
Description and references
Main bitter constituent and medicinally active principle isolated from the leaves of andrographis, q.v. Isoln: M. K. Gorter, Rec. Trav. Chim. 30, 151 (1911). Rapid isoln method: M. Rajani et al., Pharm. Biol. 38, 204 (2000). Characterization: D. Chakravarti, R. N. Chakravarti, J. Chem. Soc. 1952, 1697; R. Schwyzer et al., Helv. Chim. Acta 34, 652 (1951). Structure: M. P. Cava, B. Weinstein, Chem. Ind. (London) 1959, 851. Revised structure: M. P. Cava et al., ibid. 1963, 167; M. P. Cava et al., Tetrahedron 21, 2617 (1965). X-ray crystallographic analysis: T. Fujita et al., Chem. Pharm. Bull. 32, 2117 (1984). HPLC determn in plant material: A. Sharma et al., Phytochem. Anal. 3, 129 (1992). Determn in Chinese medicinal prepns: Z. Yanfang et al., J. Pharm. Biomed. Anal. 40, 157 (2006). Mechanism of anti-inflammatory action: Y.-F. Xia et al., J. Immunol. 173, 4207 (2004). Clinical pharmacokinetics and bioavailability from herbal preparations: A. Panossian et al., Phytomedicine 7, 351 (2000).