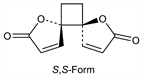
642. Anemonin
Nomenclature
CAS number: 508-44-1
(5R,6R)-rel-1,7-Dioxadispiro[4.0.4.2]dodeca-3,9-diene-2,8-dione; β,β′-1,2-dihydroxy-1,2-cyclobutanediacrylic acid di-γ-lactone; Anemone camphor; Pulsatilla camphor. C10H8O4; mol wt 192.17.
C 62.50%, H 4.20%, O 33.30%.
Description and references
Found in Anemone pulsatilla L. and other Ranunculaceae. Its precursor in plants is protoanemonin. Isoln from Ranunculus acer: Zecher, Wohlmuth, Sci. Pharm. 22, 95 (1954); C.A. 48, 13169b (1954). Structure: Moriarty et al., J. Am. Chem. Soc. 87, 3251 (1965) DOI; Romain, Diss. Abstr. B 27, 3867 (1967). Synthesis: Sugiyama et al., C.A. 67, 116604n (1967). Toxicity study: R. Brodersen, A. Kjaer, Acta Pharmacol. Toxicol. 2, 109 (1946).